
Biology
Biology Lab: AP Investigation #8 - Basic Bacterial Lab Techniques

Lab Exercise
Preparing Bacteria Cultures
Be sure that you clear this lab with your parents first.
Preparation for Lab
Before starting this lab, you may find this exercise in bacteria identification useful. Use the Bacterial Identification Virtual Lab simulation at the BioInteractive Bacterial Identification Virtual Lab website to simulate the processes required for bacterial identification. [You may need to download and configure a browser plugin for this exercise.]
Preparation:
- Click on the Bacterial Identification Lab Link above.
- Download the Worksheet (under the Materials list on the upper right), and print it off. You will use this to record your observations
- Click on "Start Interactive".
- Read through the introduction tab. You may use the updated version if you are on a tablet.
- Click on the lab picture to enter the Bacterial ID lab.
- Follow the instructions to isolate a blood sample and extract the bacterial DNA.
- Clone the DNA using a PCR method.
- Use gel electrophoresis to sequence the DNA.
- Analyze your data and identify your results.
AP Lab #8: Bacterial Transformation
AP Exam preparation -- Doing the AP version of this lab:
Please read through the 2012 AP Biology Lab Manual, Investigation #8: Biotechnology: Bacterial Transformation. This lab presents problems for home students in acquiring plasmids and E. coli strains, so most students will need to perform the alternative lab below. However, if you are able to arrange to perform the AP lab with a local homeschool group or a biology tutor, then you should do so.
Alternative Lab #1
Goal: Culture bacteria and study antibiotic sensitivities
Note that you have two weeks to complete the sections of this lab, since the first set of lab procedures is necessary to provide the bacteria cultures for the antibiotic sensitive experiments.
If you are unable to complete AP Investigation #8 as written, then perform the Illustrated Guide to Home Biology Experiments,
- Lab VII-2 Procedure 1 Preparing Cultural Media
- Lab VII-3 Procedures 1-3 Culturing Bacteria, to gain practical experience in techniques coping with bacterial investigations.
- Lab VII-4 Investigating Bacterial Antibiotic Sensitivity, Procedurs 1-3.
Be sure to follow all safety rules for handling live cultures. Complete and include in your final report any questions including in the lab instructions.
Alternative Lab #2 - Part A: Culturing Bacteria Strains
Goal: Culture bacteria and study antibiotic sensitivities using household food preservatives
Note that you have two weeks to complete the sections of this lab, since the first set of lab procedures is necessary to provide the bacteria cultures for the spice sensitive experiments.
Goal: To grow different bacteria cultures in media. Although we will be using bacteria which is already part of your household environment, you should treat all the instruments and cultures as though they were contaminated by serious pathogens.
Materials:
- A medium (environment with food) for the bacteria.
- The most often used medium is an enriched agar. You can buy agar (a protein made from sea kelp) from Japanese grocery stores and from science supply houses.
- Unflavored gelatin mixed with beef extract. You can get unflavored gelatin at the grocery store. You can make beef extract from leftover beef (boil bones and bits of meat to get a thick stock). Be careful if you substitute boullion or stock--it often has a lot of salt, which will inhibit bacterial growth. Don't use jello--it has too much sugar for the bacteria to grow!
- An inoculation loop
- A flame resistant loop can be made from a piece of NiCad wire, bent into a small loop shaped like this: ----o The loop should be firmly inserted in the tip of a wooden dowel.
- A non-flame resistant loop, which can be (less effectively) sterilized with isopropyl alcohol, can be made by bending a paper clip into the desired shape and inserting the wire tip into a dowel.
- If all else fails, use separate Q-tips for each inoculation.
- Petri dishes, or you can use baby food jars or any other shallow heat-proof glass jars, like canning jars. You will need one pair of flats for each bacteria source you use; and you should do a minimum of three sources (i.e., 6 flats).
- You can get a vial of agar, inoculation loop, and 3 pre-sterilized petri dishes, plus instructions, in a nice little kit from Science and Discovery series for about $7. These kits come in sandwich size "baggies" and are sold at educational toy stores and science museums, or you can check my suppliers page for their address.
If you prefer, you can order a kit with all of the above components from Ward Science.
Procedure
Creating the colony.
- Sterilize your "flats". [You can skip this step if you have sterile Petri dishes to start with]. Wash your glassware in hot, soapy water. Rinse all the soap off with hot water, then pour boiling water inside, dump, and turn upside down. Pour boiling water over the outside, and let air dry. Use appropriate caution when working with boiling water, and remember the first rule of the chem lab: hot glass looks like cold glass, so pay attention to what you are doing and wait for the glass to cool before handling it, or use canning tongs. Do not touch the inside of the jars after this point, and leave upside down or covered unless working with the jars.
- Mix up the medium. Follow the directions for agar or unflavored gelatin by adding hot water to dissolve the dry medium, then letting the mix cool to make a stiff "jello" like substance. If your mixture is still runny when at room temperature, cook it some more and let it cool again.
- Warm the medium gently and pour it into your "flats" so that it forms a thin, transparent layer across the bottom of the flat.
- Sterilize the inoculation loop appropriately (or use a new Q-tip for each inoculation).
- "Infect" the loop by exposing it to a bacteria source, for example:
- the back of your ear
- the inside of your cheek (cavies bacteria that cause tooth decay)
- the sweat between your toes
- garden soil
- soured milk (leave small amount standing in open air for 20 minutes, then cover and let sour for 24 hours)
- ????
- Take one of your flats and streak the top surface gently (don't break the surface!) with the inoculation loop or Q-tip. Use one of the following patterns:
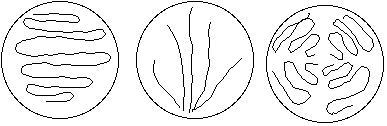
Bacterial Plate Smear patterns
- Mark the flat with a crayon so that you can identify the source of the bacteria and the streak pattern you used.
- Cover the flat and leave it in a room-temperature area out of direct sunlight AND AWAY FROM YOUR NORMAL FOOD SUPPLIES!. Check it daily for one to two weeks (the amount of time necessary to achieve a colony will depend on temperature, the concentration of the agar, and similar conditions), and record the changes in appearance.
- Repeat the inoculation procedure for at least two more bacteria sources (three altogether as a minumum, six-ten are better if you have the media and flats to do it).
- If possible, create two sets of flats (complete exposures from all your sources). Place them in different environments (both dark, but one colder than the other).
Counting the colonies
- When you have some thriving cultures, you need to count them. Place a piece of graph paper under the dish so that the graph "squares" or "cells" show through the medium.
- Count the number of "colonies" in each square. A colony of bacteria look like a round, moist drop of colored stuff. You may also get fungi from your exposure of the media to different sources. You can tell the difference because fungi have a threadlike appearance like a flat fuzzy ball.
- Describe the appearance of each colony (large, small, individual dots, colors, texture).
Alternate Lab #2 - Part B: Spice Sensitivity
Goal: To observe changes in the response of bacteria to preservatives, such as spices
Materials
- Bacterial cultures (from Part A)
- Spices such as black and cayenne or chili pepper, cinnamon, salt, cloves, vinegar
- Q-tip or eyedropper
- Small jars or cups to make spice concentrations.
Procedure:
- Examine your flats and record their appearance. If necessary, make a drawing to show the location of the bacteria colonies and add a description of their color and type.
- Chose a number of spices equal to the number of bacteria flats that you have (i.e., if you have 5 bacteria flats, choose five different spices)
- Make an infusion of each spice by adding 1/4 teaspoon of the ground or crushed spice to 1/4 cup of boiling water. Be sure to record the amounts you actually use.
- Let the infusions return to room temperature.
- Using a Q-tip or eyedropper, add a small amount of each spice to several spots on each flat. Be sure to leave some healthy colonies unexposed to the spice. Record the location of your spice additions (you may want to make a grid drawing and place it underneath the flat so that you can tell later where you put the spice infusions).
- Observe your flats for 3 days. Record the appearance and note any changes in the bacteria cultures, both near and far from the spice additions.
- Summarize your findings: which spices were effective in destroying or limiting bacteria cultures? If you had more than one type of bacteria culture, which seemed most sensitive to which spices? If you had fungus (thread-like growths as opposed to round bumpy colonies), did they respond to the spice infusions?
- You should dispose of your colonies now. In order to minimize the danger, you should flush all colonies and media down the toilet (which is already handling a serious amount of E. coli, and can probably handle anything you would find around the house). Use a sink other than one where you would prepare food, and wash your glassware with hot, soapy water. Rinse thoroughly, then rinse again in a bleach solution (1 cap to gallon of water). Let air dry complete before storing in a dry place or discarding.
Report
- Follow the usual practice of stating your goals, outlining your procedure and methods, with particular detail if you deviated from the procedure above.
- What patterns do you notice in bacteria growth? Do bacteria colonies from similar sources look alike?
- If you placed flats in different areas, how did differences in light, humidity, and temperature affect the growth of the colonies?
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.