
Chemical History of the Candle
Lab Assignment 5: Splitting Water

LAB: Composition of Water
Goal: To separate water into hydrogen and oxygen gas
Materials:

- Two insulated light-weight wires. If possible, use stainless steel wrapped in electrical tape (with the ends uncovered), rather than copper wires.
- 6-Volt battery (or 4 AA 1.5 V batteries taped in sequence)
- Large beaker or glass canning jar (like a large peanut butter jar)
- Two test tubes or small spice or medicine jars which will fit upside down together in your beaker
- Water
- Epsom salts or baking soda (not table salt).
- Wood flints or used wooden match sticks.
- Candle or other flame source.
Procedure:
- Mix a tablespoon or so of epsom salts or baking soda into a cup of distilled water. We use "salted" water because it conducts electricity better than plain water. [You could this experiment with pure distilled water but it would take a lot longer to get any appreciable amount of gas.] Table salt will work as an electrolyte, but the released sodium will also interact with any oxygen produced. If you use salt and copper wires, you will get hydrogen, but not oxygen.
- Leave about 1/2 of the end of each wire free of insulation.
- Assemble your apparatus as shown. Note that as the tubes collect gas,
the water in them will be forced out into the beaker and may overflow if
the beaker is already too full.
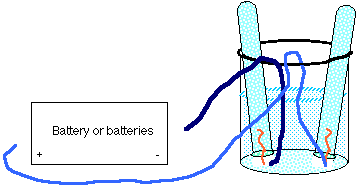
Battery not yet connected (step 3)
- Be sure that the test tubes are completely full of saltwater in their inverted position, so that the gas which collects in them is not contaminated with air. You may need to practice to get them into the water bath and then get the wires into position.
- Mark the tube which will be connected to the postive terminal with a +.
- Mark the tube which will be connected to the negative terminal with a -.
- Note the time.
- Connect the wires to your battery and allow gas collection to occur
in the test tubes.

Battery is connected (step 8)
- Allow your collector to run until both tubes have a significant amount of gas in them (about half full in one of the tubes -- the other may have less).
- Without removing the tubes entirely from the water, mark the level of gas in each tube.
- Now identify the gases in each tube:
- Carefully remove one of the test tubes from the water, slipping your thumb over the end of the test tube before it leaves the water (it should have both water and gas in it).
- Light your flint from the candle and blow it out so that it is lightly smoldering.
- You have to do this part quickly----you may want to practice with "empty"
tubes first.
- Move your thumb aside and quickly place the flint about two inches above the tube. Record your observations.
- Quickly cover the open end of the tube again, invert it so that the water runs out, and place a glowing flint inside. Record your observations.
- Repeat the tests with the other tube.
- Empty the test tubes and measure the part of the tube from the bottom (closed end) to your mark for the level of the gas. Record the length for each tube.
- Possible variations
- Repeat the experiment with plain water and with double the salt you originally used, and compare the amount of time required to generate the same amount of gases.
- Use other substances in the water (sugar, baking soda, vinegar). Which works best?
Report:
- As usual, describe your procedure and any special materials you used.
- Based on your observations with the flint, which tube contained hydrogen and which contained oxygen? Why?
- Compare the two amounts of gas produced. Which gas occured in the greater amount? How much more was produced? What conclusions can you draw about the composition of water from this experiment?
If you were to mix the two gases in a common vessel and run an electric spark through them (DON'T DO THIS--IT IS DANGEROUS without the proper equipment!) you would get water again, along with A LOT of energy in the form of light and heat. The powerful reaction of liquid oxygen burning hydrogen gas is what drives the Space Shuttle skyward--and produces, as "waste" from the reaction, only water.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.