
Chemical History of the Candle
Lab Assignment 3: Measuring Oxygen in the Atmosphere

LAB: Resources for Combustion
Goal: Measure the amount of atmosphere consumed by a burning candle
Materials:
- A short candle with a good wick.
- A deep pan or bowl in which you can put at least a quart of water.
- A heat proof jar (such as a canning jar) or chemical beaker.
- Matches
- A wax marker like a crayon
- A liquid measure, graduated to as much accuracy as possible (a cup measure will do, a graduated cylinder is better).
Procedure
- Melt a little wax into the pan and fix your candle into it so that it won't fall over.
- DON'T light the candle yet.
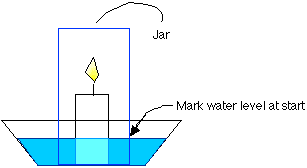
- Fill the bowl with water, but make sure that at least 1 inch of candle stands above the water line.
- Upend your jar over the candle and mark the position of the water against the jar at the start of the experiment.
- Take the jar out.
- NOW light the candle.
- Quickly upend the jar and put it back into position.
- Wait until the candle flame dies completely.
- WITHOUT MOVING THE JAR, mark the new position of the water line.
- Take the jar out of the bowl
- Fill the jar to the first mark (this should be the mark after the candle burned out) with water.
- Pour the water into your measuring cup or graduated cylinder and record how much water you had. This is the volume of air left after the flame exhausted the whatever-it-is in air that keeps things burning (yes, we know it is oxygen, but we're trying to be Lavoisier at the moment....)
- Now fill the jar to the second mark (this should be the original starting mark) with water
- Pour the water into your measuring cup or graduated cylinder and record the amount. This is the volume of air you started with.
- Divide the left-over amount by the original amount: this is the non-combustible part of the air.
- Repeat the experiment for a total of three measurements and average your results.
Report
Describe your setup and procedures. Record your measurements in a nice tabular form:
| Trial # | Left-over volume | Starting volume | Left-over/starting |
| 1 | |||
| 2 | |||
| 3 | |||
| -- | -- | AVERAGE → |
Be sure to address the following questions in your conclusions:
- What percentage of the atmosphere is a gas that supports combustion?
- Why do we repeat the experiment?
- Why do we average our results?
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.