
Lab Assignment: Density and Experimental Error

Forces of Nature Lab
Density, Measurement, and Error
Goal: To measure the density of an regular solid.
[This is a modification of the AP Physics Lab Making and Calibrating Lab Equipment. If you intend to submit a lab for AP credit in the Fall Scholars Online Advanced Placement/Honors Physics course, you should the instructions at that site.]This requires that you measure two properties directly (mass and volume), then compute the density as mass/volume. You will need to obtain or construct an equal-arm balance capable of measuring 1 gram differences.
How you build your scales depend largely on the materials you have available. BE SURE THAT YOUR PARENTS AGREE TO YOUR USE OF ALL MATERIALS! The possibilities suggested should stimulate your imagination: improve on them and don't limit yourself to them.
Materials and Procedure
Equal arm scales work by comparing one mass directly to another. When the scale is "balanced" and not moving, all gravitational forces sum to zero, so the only factor left is the mass itself.
You will need
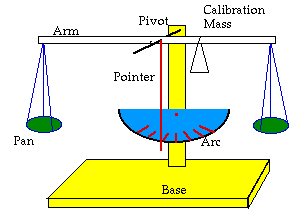
- the arm: a straight length of wood or metal, lightweight and rigid. Possibilities: dowel, yardstick.
- a pivot mechanism: this will take the weight of the arm at the center; the arm must move around this point. Possibilities: suspend the arm on a needle/nail passed through a hole at the center and near the top edge of the arm, or set the arm on top of a narrow ridge, such as a needle or razor-blade.
- a support for the pivot: this keeps your pivot from moving and puts it high enough that your arm and pans swing freely above the table. Possibilities: a used milk carton of which you can fasten or embed your nail/needle pivot; a Styrofoam cup which you can put on top of a stack of books. Put something heavy in the bottom of the carton or cup to keep it from tipping over.
- a calibration weight: this should be something on one arm which you can slide along the arm to make the balance read zero when your pans are empty. It will compensate for irregularities in weight of the arm and pans. Possibilities: a bent paper clip from which you can suspend rubber bands or other small counterweights.
- weighing pans: these are lightweight containers fastened to the each end of the arm. In one you will place calibrated weights, in the other you will place the object(s) to be weighed. Possibilities: flat lids, suspended from the ends by thread, fishing line or wire; paper cones or cups tacked to the ends of the arms.
- pointer: this extends at right angles from the pivot point. When it is vertical, your pans are balanced. It needs to be lightweight and rigid. Possibilities: coffee stirring straw, small wooden skewer, fastened at aright angle to the arm at the pivot point.
- calibrated arc: this allows you to measure how far from true vertical your pointer is. You will need to place the arc (which should describe part of the circle made by the pointer when the scale is oscillating on its pivot) behind the pointer. How can you center the arc properly?
- weights: These are calibrated masses which you will use to counter the mass of the objects you measure. You will need to find or make masses to measure .1, 1 and 5 grams. Possibilities: a 1990 or later nickel weighs 5 grams when newly minted. How can you make a set of 1 gram standards? Some medicine tablets are pure substance: 500 mg of pure aspirin makes a good .5 g mass.
Data Handling: Derived quantity measurement
Now you need to determine the volume of a regular object. Use some other cube-shaped object which has a mass about that of 5 nickels. If you don't have a good candidate, choose some medium (I like using firm potatoes) and cut a cube 3 cm per side (if exact, your total volume will be 27 cm3). Measure the sides and estimate the error in each of your measurements--remember that these may be due to irregularities in the cube as well as in your measurements. Calculate the volume of your cube and the possible error in volume.
Now weigh the cube, using one or both scales. Take at least three different measurements of the mass (remove and return the mass to the scale). If possible, have someone else perform a mass measurement. Determine the average mass measurement (sum all the masses measured and divide by the number of times you performed the measurement). Estimate the error in your mass measurements.
Calculate the density (mass/volume) of your cube, using your average measurements and your extreme measurement possibilities (greatest amount of error more and less). Determine the error as a percentage of the average density derived from the separate measurements.
Report
- Describe your materials, equipment, and procedures in sufficient detail that mycroft, our invaluable lab assistant, could repeat your experiment.
- Report your data. Be sure to indicate the amount of error in your measurements. For example, if you can only measure a mass of 25gm within 1 gm, your error would be 25 ± 1, or 1/25 = 4%.
- Present your data in an organized form, preferably in a table, in such a way it is easy to compare results as you repeat trials or vary a specific contributing factor.
- Show a sample calculation, if you have calculated values.
- If you did a series of experiments, varying something by increasing or decreasing a factor, try to plot your data (y-axis) as a function of the factor (x-axis).
- You may use a spreadsheet to calculate your information and create your table.
- Summarize your results.
- Draw conclusions about what is happening.
- Suggest at least one way to improve your experiment.
- Post your results to the Moodle forum for this course.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.