
Physics
Chapter 16: 1-6

Web Lecture
Electrical Charge
And we daily in our experiments electrise bodies plus or minus, as we think proper. [These terms we may use till your Philosophers give us better.] To electrise plus or minus, no more needs to be known than this, that the parts of the Tube or Sphere, that are rubb'd, do, in the Instant of Friction, attract the Electrical Fire, and therefore take it from the Thin rubbing; the same parts immediately, as the Friction upon them ceases, are disposed to give the fire they have received, to any Body that has less.
— Benjamin Franklin Letter 25 May 1747.
Outline
A History of Theories on Electric Charge and Static Fields
Charge is the property of matter that gives rise to electrical force, just as mass gives rise to gravitational force. The two are very similar in some ways (compare Coulomb's law for electrical force with the form of Newton's Universal Law of Gravity), but unlike mass, which is always positive, charge comes in two kinds, positive and negative, which have different effects on each other.
Theories from the Ancient World to the Enlightenment
We begin our detailed investigation of electricity and magnetism by looking at the phenomena associated with static (unmoving) charges. These phenomena are all around us--every time you "zap" yourself by walking across a dry rug and touching a metal doorknob, or try to get your hair to lie down while combing it on a dry day, or when you watch a lightning bolt flash across the sky, you are looking at static electricity in action. From ancient times we have many records of observations of electrical phenomena, from lightning and St. Elmo's fire to electrical eels, and the attractive power of rubbed amber. Thales of Miletus is reported to have talked about how rubbing amber stone with cat fur made it spark. The amber didn't have to be in direct contact with the fur after it had been "charged" by rubbing; just bringing the fur close to the amber would make it glow. Obviously, something was influencing the behavior over the intervening space.
Plato made a list of such phenomena in his Timaeus:
Moreover, as to the flowing of water, the fall of the thunderbolt, and the marvels that are observed about the attraction of amber and the Herculean stones, — in none of these cases is there any attraction; but he who investigates rightly, will find that such wonderful phenomena are attributable to the combination of certain conditions — the non-existence of a vacuum, the fact that objects push one another round, and that they change places, passing severally into their proper positions as they are divided or combined.
Although he was mistaken about the attraction (there is an attractive force involved), Plato's list suggests that not only were people aware of electrical phenomena, but that they also recognized similarities between this class of events.
There are also ancient and medieval records of magnetic effects, which were often interpreted as magical or at the very least, partaking of the deepest mysteries of nature. St. Augustine in Book XXI of The City of God gives an excellent description of the attraction of lodestone for iron, and the ability of diamonds to block this attraction, based on his own observations. His purpose is to show how phenomena that cannot be accounted for by reason may nevertheless be true, but the account demonstrates that certain magnetic and electrical phenomena were well known, however mysterious. Magnetism was the subject of investigation by the English scientist William Gilbert, who published his book De Magnete in 1600. Gilbert discussed both the behavior of magnets and different electrical phenomena, even listing a number of objects subject to static charges like the amber and cat fur. But along many of his contemporaries, he believed that magnetism and electricity were unrelated phenomena, and his theory influenced philosophers for the next two centuries.
In the late seventeenth century, the German Otto von Guericke used a more mechanical approach to investigating static electrical phenomena. Guericke invented a vacuum pump, but his research was not confined to the properties of vacua and air pressure. He build a machine to generate electricity (a ball of sulfur which rotated against a rough surface, absorbing charge from it), and showed that the resulting charges could repel as well as attract different objects. You can duplicate von Guericke's experiment with an inflated balloon: just rub it against your hair or a wool sweater on a dry day, and you'll create enough charge on the balloon that electrical attraction will hold it to a wall and keep it from falling under the force of gravity.
Fifty years later, Stephen Grey sent a paper to the Royal Society in which he explained how he transmitted charge from a generator like von Guericke's over 650 feet in metal wires suspended by silk thread. He discovered that metal suspension wires wouldn't work, since they acted as grounds and transmitted the flowing charge back to earth before it could reach the end of the wire. He thus gave the first descriptions of materials as insulators (non-conductors) and conductors of electricity. He also discovered that he could induce a charge into an object merely by bringing another charged object close to the first, then removing the first from contact with the ground. Grey's report is the first evidence of someone inducing electrical charge.
Charles du Fay, a French scientist, repeated Grey's experiments with the same results—a valuable confirmation, since du Fay's experiments were done in a different country with different equipment. Du Fay also proposed that there were two different electricities, vitreous and resinous. Vitreous electricity in one object repelled vitreous electricity in another object, but attracted resinous electricity.
Among others (the invention is usually attributed to Pieter van Musschenbroek in 1745, du Fay learned how to "store" charge in a Leyden jar, a glass container with metal inserts which did not quite touch. The Leyden jar was the first capacitor. This was an important invention, because it allowed experimenters to keep charge around for experimenting at their own convenience; they no longer had to wait for a nice dry day when collection would be easy.
Another major step was taken by the American colonial Benjamin Franklin, who became fascinated with electrical phenomena and turned his considerable intelligence and disciplined industry on the problem. He came to the conclusion that lightning is electrical in nature, which he attempted to prove by flying a kite during an electrical storm. Just being in the thundercloud was enough; the kite collected charge, sent it down the wet string and into the Leyden jar which Franklin was using. Contrary to some versions of the story, Franklin's kite was not hit by lightning, but that of a young man who tried to repeat Franklin's experiment was, and the man was killed [so do not try this experiment yourself]. Franklin described his research in a series of letters which he sent to Peter Colinson, then president of the Royal Society, who arranged for their publication in England.
Read the description of Franklin's "lightning bells" at the Franklin Institute, the science museum in Philadelphia which Franklin started.
- What was the purpose of Franklin's bells?
- Was Franklin able to make any measurements of the electrical charge involved in ringing the bells?
- What conclusions did he draw from his observations?
Franklin's studies led him to the conclusion that electrification of an object did not create charge in the object, but moved it there from somewhere else. He was the first to voice a law of the conservation of electrical charge, which joined Lavoisier's rule about the conservation of matter as two of the three great conservation laws of physical science (the third is the conservation of energy).
Joseph Priestley also studied the affects of electric attraction over distance and realized that the attraction of electricity was subject to the same distance law as gravity, that is, the force exerted by an electrical charge varied as 1/r2.
The man who put all these observations together was the French scientist Charles-Augustin de Coulomb. He continued Priestley's experiments, and in 1785, published the rule that
Force due to electricity = q1 * q2 * k /r2,
where q1 and q2 where two different sources of charge and r was the distance between them. He knew that k was a constant of proportionality, like the factor G in Newton's equation for the force of gravity, and eventually it was determined to be 8.99 * 109 N*m2 /C2. N stands for Newtons, the force unit, m for meters, and C is Coulombs, the units of charge.
Charge — the modern approach
All of our attempts to account for charge reduce to the fundamental atomic particles: protons and electrons. In any given atom, the protons in the nucleus carry one positive charge apiece, the electrons one negative charge apiece. It is the attraction of electrons for the positively-charged protons in the nucleus of their own atom that holds the partos of the atom together. It is the attraction fo electrons for any positive charge — including the protons in the nucleus of another atom — that bond atoms to one another to form molecules.
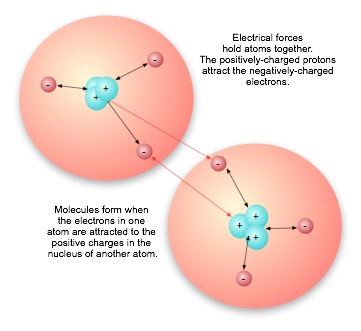
Even when the net charge in a structure is zero, local charges can create dipoles, concentrations of opposite charges, that then attract other oppositely-charged dipole areas. An exceedingly important example of polar molecules is the water molecule. The highly positive nucleus of the oxygen atom keeps not only its own electrons, but the single electron of each hydrogen atom in the water molecule, close to the oxygen nucleus. This leaves the proton of the hydrogen atoms unshielded, so that it appears to other atoms as a local area of positive charge. The attraction is much less than the attraction of the hydrogen for its own electron, but it is enough to raise the boiling point of water significantly.
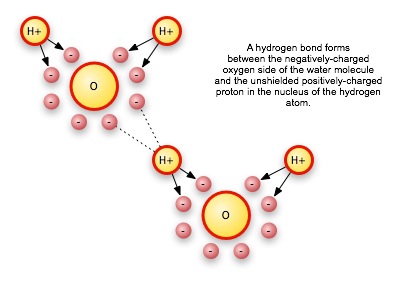
Under some circumstances, the negative charge on one water molecule can even pull a hydrogen off of another molecule, resulting in two ions, or charged molecules.
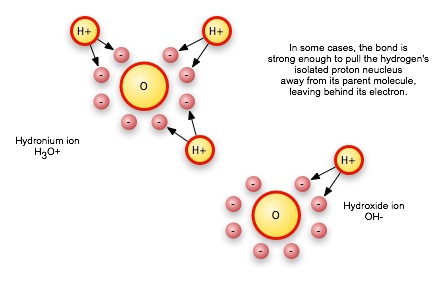
It is this last phenomenon, the flow of electrons from one place to another, that accounts for most changes in local charge and allows a given area to accumulate a net positive or negative charge.
Static Electricity
When we consider the forces that arise from electrical charge, it is useful to begin with a review of gravity. Both mass and charge are properties of matter, each can exert force across distance without necessarily any intervening medium, and both depend on conservation laws.
Gravity is a property of all kinds of matter. Anything with mass exhibits an attractive force toward anything else with mass--an interesting quantity which is considered constant for the universe as a whole (matter cannot be created or destroyed). The magnitude of gravitational force is a product of the masses involved and depends on the inverse square of the distance between them.
Electrical charge is also a property of all kinds of matter. Since it may be either repulsive or attractive, its effects in one object may cancel, leaving the object apparently neutral. The total charge in the universe, like the total matter and energy, is considered constant. Charge can be neither created nor destroyed.
It can, however, be transferred. According to the atomic model of electrical charge, two sub-particles of the atom carry electrical charges, equal in magnitude, but opposite in effect. The proton, which is usually part of the atomic nucleus, carries a positive charge. The electron, which exists in a fixed area around the nucleus (depending on the electron's energy), carries a negative charge. When two objects are rubbed together, electrons may be pulled off of some atoms and attach to others, creating an electrical charge imbalance on both objects. The resulting charge is called static electricity, because the charge is not moving through the object.
If we bring a charged object (one which has either lost electrons and become positive or gained electrons and become negative) in contact with an uncharged object, electrons may move from one object to the other. Like heat flow, the electrons will move in whichever direction will reduce the overall charge difference.
Not all objects give up or receive electrons with equal ease. Two metal spheres made of the same material would charge equally well. If we charge one and let it touch the other uncharged sphere, both spheres will become charged. However, it is possible to place a piece of wood between the two spheres, and even though a charged sphere touches the wood on one side and the uncharged sphere touches the wood on the other side, no electrons move and the uncharged sphere remains uncharged. Materials which are reluctant to accept or move charges are insulators; materials in which charges may move freely are conductors. Most metals are conductors, although some, which are noticeably less efficient, are called semi-conductors.
Detection of static electrical charge can be detected and measured using electroscopes or electrometers. For a discussion on how to build and use one, see this week's lab.
As we already saw, Charles Augustin Coulomb is credited with the formulation of the rule relating electrical force with charge. After a number of experiments and careful measurements, he concluded that the force due to electrical charge between two objects is proportional to the product of the charges involved and the inverse square of the distance between them:
The value of k depends on the units used for charge and distance. If charge is in coulombs and distance is in meters, than k = 8.988 *109 Newton-meters2/Coulomb2. This constant of proportionality mas also be written in terms of the permittivity of free space, which is a measure of the ability of a non-conducting space to transmit electrical force. The permittivity constant is usually written using ε0 = 1 / 4 * π *k = 8.85 * 10-12 Coulomb2/Newton*meter2.
We use this law the same way that we used Newton's law of gravity. There, we assumed that the force exerted by mass M could be determined by placing a test mass m within the gravitational influence of M.
In the same way, most of our applications for Coulomb's law involve placing a test charge q1 = q within the electrical influence of a given charge q2 = Q:
Practice with the Concepts
Discussion Points
- If you charge a pocket comb by rubbing it with a silk scarf, how can you determine if the comb is positively or negatively charged?
- Explain why fog or rain droplets tend to form around ions or electrons in the air.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.