
Physics
Chapter 10: 1-7

Web Lecture
Principles of Fluid Mechanics
Introduction
On Floating Bodies
Let it be granted that the fluid is of such a nature that of the parts of it which are at the same level and adjacent to one another that which is pressed the less is pushed away by that which is pressed the more, and that each of its parts is pressed by the fluid which is vertically above it, if the fluid is not shut up in anything and is not compressed by anything else.
Archimedes of Samos, On Floating Bodies Book II
Outline
From chapters 1-9, we've spent all our time looking at how rigid bodies or solids move when forces act on them. We know that a force through the center of mass causes the body to change position (translational motion) and forces through any other point will cause the body to rotate around a fixed point (or around the center of mass if there is no fixed point). In chapter 10, we look at what happens when forces are exerted on fluids, and in chapters 13 and 14, we'll look at what happens when forces (in the form of pressure are exerted and when energy changes in gases.
Bodies in Fluids
(We start by considering fluids in containers, that is fluids that are not actually flowing except around objects placed in the liquid).
Fluids consist of particles that are able to move independently of one another to some extent. As a result, exerting force on the fluid will cause it to deform rather than move with simple translational motion or rotation. We need another way to describe what is happening, one that allows us to compare forces exerted over areas (through the middle of many particles) rather than singly to a center of mass. We define pressure as force exerted over area: P = F/A.
Fluid Pressure
The response of a liquid to pressure depends on its density. We define density as mass/volume, or ρ = m/V (where capital V is often used to distinguish it from v=velocity). To get the volume of a liquid of known density, it is often easier to measure the mass directly, and calculate the volume from V = m/ ρ, rather than measure the volume, especially if the volume is irregular.
The pressure that a column of water exerts on an object at a given depth depends entirely on the gravitation force of the mass of water above the object. The water above the object occupies a volume equal to the "upward" surface of the object and the height of the water. If A is the surface area, then the pressure on the object is P = F/A = mg/A. Now since m = ρ*V and V = h*A, we can substitute into P = F/A an expression for F = mg/A = ρ*V g/A = ρ *hAg/A = ρgh = P. This gives us a number of equivalent expressions for pressure, any of which can be useful depending on what we know of a given situation.
The fact that particles in a liquid can slide around one another does not necessarily mean that they can be squeezed more closely together, such that the liquid is compressed. Water molecules in particular are polar, that is, one side of the molecule has a negative charge while the other has a positive charge. These local charges affect the orientation of nearby water molecules, repelling the sides of molecules with like charges and attracting the sides with opposite charges. Ultimately, there is a limit to how closely packed the molecules can be: they cannot merge. Water is already closely packed under normal atmospheric pressure, and even at great depths in the ocean, cannot be compressed any further.
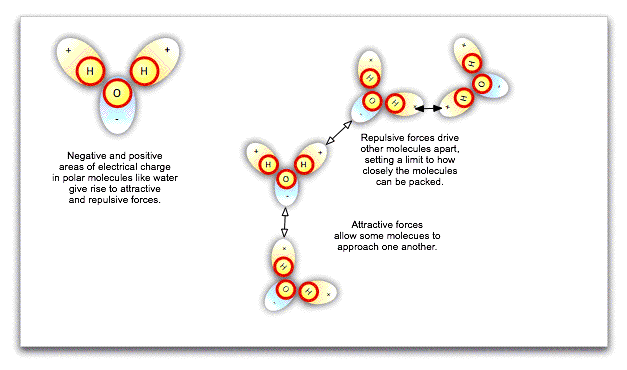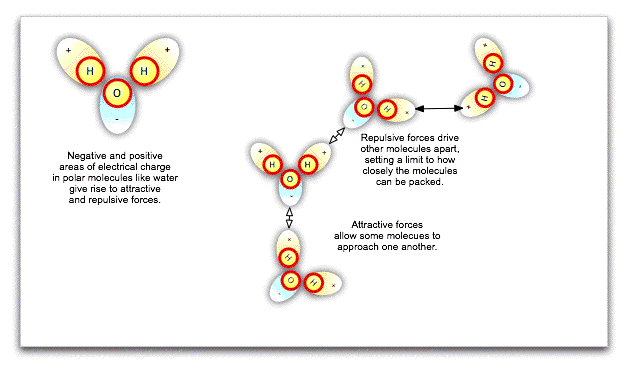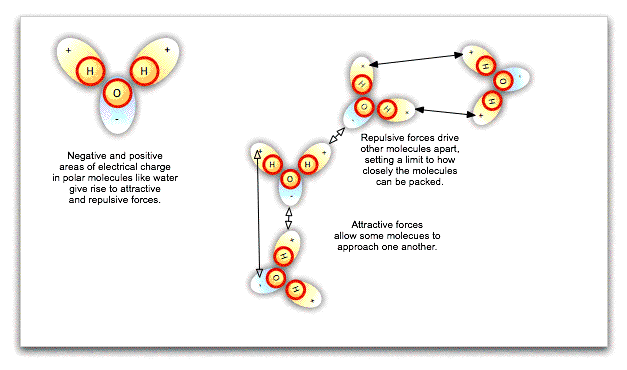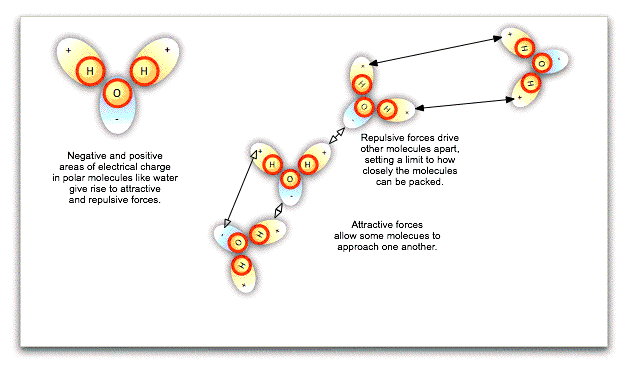
If we exert pressure on the surface of an incompressible liquid like water, the molecules will push against one another at different angles, spreading the pressure equally throughout the liquid's volume, however irregular that may be. This is Pascal's law, and it has enormous practical consequences. Hydraulic lifts at the local garage, not to mention the hydraulic brakes in your car, work because exerting small pressure on a large area of liquid can be translated into large pressure on a small area.
Units of Pressure
We have a number of units and instruments for measuring pressure. It is important to realize that most gauges, unless otherwise instructed, assume a zero point equal to the earth's atmosphere at sea level, and measure the amount of pressure above this baseline. Absolute pressure must take the baseline into account. [This is similar to the relationship between centigrade temperature, which establishes its zero point at the temperature at which water freezes, and absolute or Kelvin temperature.] If we use a tire gauge to measure 32 pounds of pressure in a tire, this is the difference between the tire pressure and atmospheric pressure. The absolute pressure of the air inside the tires is actually the gauge pressure plus the atmospheric pressure, or 32 + 14.7 = 46.7 pounds per square inch.
It may be useful here to show the advantages of using a spreadsheet with some of these calculations. We create a row (row 1) with formulae that covert 1 atm to the correct number of different units. Column A will hold the values for atmospheres. We write the formula for each succeeding column to multiple whatever value is in column A of the same row by the correct factor for the units of the column. Column B will hold the equivalent values in Pascal, that is, 1.013 * 10^5 N/m2 times the number of atmospheres in column A. Column E holds the number of pound per square inch, which is 14.7 times the number of atmospheres in column A.
Now all we have to do is put values in column A and "replicate" the values in the rest of the columns for that row, using the formulae in row 1. We can generate in a few seconds values for 1, 2, 10, 50, 100 and 1000 atmospheres.
| Col/Row | Col A | Col B | Col C | Col D | Col E | Col F | Col G | Col H | Col I | Col J |
| Row 1 | 1 | =A2 * 1.013 * 10^5 |
=A2 * 1.013 |
=A2 * 1.013 * 10^6 |
=A2 * 14.7 |
=A2 * 2.12*10^3 |
=A2 * 76 |
=A2 * 760 |
=A2 * 760 |
=A2 * 1.03 * 10^4 |
| Row 2 | ATM | Pascal (1 N/m2) |
Bar | Dyne/cm2 | lb/in2 | lb/ft2 | cm-Hg | mm-Hg | Torr | mm-H20 |
| Row 3 | 0.1 | 10130 | 0.1013 | 101300 | 1.47 | 212 | 7.6 | 76 | 76 | 1030 |
| Row 4 | 0.5 | 50650 | 0.5065 | 506500 | 7.35 | 1060 | 38 | 380 | 380 | 5150 |
| Row 5 | 1 | 101300 | 1.013 | 1013000 | 14.7 | 2120 | 76 | 760 | 760 | 10300 |
| Row 6 | 2 | 202600 | 2.026 | 2026000 | 29.4 | 4240 | 152 | 1520 | 1520 | 20600 |
| Row 7 | 10 | 1013000 | 10.13 | 10130000 | 147 | 21200 | 760 | 7600 | 7600 | 103000 |
| Row 8 | 50 | 5065000 | 50.65 | 50650000 | 735 | 106000 | 3800 | 38000 | 38000 | 515000 |
| Row 9 | 100 | 10130000 | 101.3 | 101300000 | 1470 | 212000 | 7600 | 76000 | 76000 | 1030000 |
| Row 10 | 1000 | 101300000 | 1013 | 1013000000 | 14700 | 2120000 | 76000 | 760000 | 760000 | 10300000 |
Practice with the Concepts
Discussion Points
- A dam holds back a lake that is 85m deep at the dam. If the lake is2 0km long, how much thicker should the dam be than if the lake were smaller, only 1.0 km long?
- Which experiences the largest buoyant force: A 1-kg helium balloon; a 1-kg block of wood, a 1-kg block of ice? Or do all of them experience the same buoyant force? What happens if, instead of having equal mass, each object occupies the same volume?
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.