
Chemistry
Calorimetry

Laboratory Exercise
Goal: To determine the heat capacity of a simple calorimeter.
Materials:
- Clean 2 qt pot
- Large beaker or glass jar capable of holding the styrofoam cups (for stability).
- Two styrofoam cups, same size, at least 8 oz (1 cup) capacity.
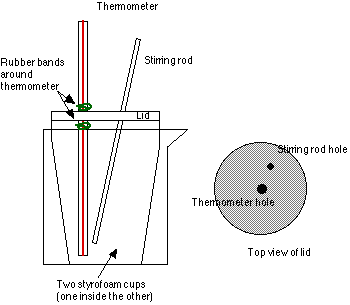
- Lid for styrofoam cup. You can make a lid out of aluminum foil, paper, or cut a sheet of styrofoam or cardboard to fit. The lid should have two small holes, one for the thermometer, one for the stirring rod.
- Thermometer, with a range of 0 to 110 °C or 20 to 220 °F [Two if you have them]
- Glass stirring rod. You can substitute a wooden chopstick or skewer. but don't use metal. Why not?
Procedure
- Nest the two cups together and set them inside the beaker.
- Place the thermometer in the lid, and position it so that when the lid is on the cup, the thermometer is 2-3 cm above the bottom of the cup, and not touching the sides. You can secure the thermometer in place by wrapping a rubber band around it above and below where it passes through the lid. Remove lid.
- Measure 100 mL of water and place it in the inner styrofoam cup. This is mo (original amount of water). If you use a different amount, record the amount.
- Place the lid on the cup, and wait until the thermometer stops changing. Record the temperature of the water. This is To.
- Using the pot, bring 100-200mL of water to a boil. Measure 30 mL into a small beaker. If you use a different amount, record the amount as ma (Water added).
- OPTIONAL: If you have a second thermometer, measure the temperature of the water in the small beaker. Otherwise, assume that it is 100 °C. This is Ta.
- Add the 30mL of boiling water to the styrofoam cup calorimeter, reseat the lid, and use the stirring rod to stir the water. Try not to hit the thermometer.
- Watch the thermometer. Record the highest temperature attained. This is Tf.
To calculate the heat capacity of the calorimeter, you calculate the change in temperature of the hot water (Ta - Tf = Taf) and the change in temperature of the cold water (To - Tf = Tof ).
The calorimeter constant is given by -C(mo*Taf + ma*Tof) / Taf. The heat capacity C of water is approximately (1 cal/g-K).
Report:
Your lab report should include a description of the materials and equipment you actually used, your procedure, all data collected, and the determination of your calorimeter constant for at least two trials using different amounts of water in the same equipment.
Don't throw away your equipment! You will need it for the next experiment.
You might also find the Calories Count lab interesting.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.