
Natural Science - Year II
Unit 55: Rutherford and Atomic Structure

History Weblecture for Unit 55
| This Unit's | Homework Page | History Lecture | Science Lecture | Lab | Parents' Notes |
History Lecture for Unit 55: Atomic Structure and Atomic Bonds
For Class
- Period: 1850-1930
For the interactive timelines, click on an image to bring it into focus and read notes.
Click on the icon to bring up the timeline in a separate browser window. You can then resize the window to make it easier to read the information.Click here: Timeline PDF to bring up the timeline as a PDF document. You can then click on the individual events to see more information if you want. Exploring this version of the timeline is optional!
- Geographic Location: England, Denmark!
- People to know: Niels Bohr, Ernst Rutherford, Max Planck, Albert Einstein, W. Schrödinger, Linus Pauling
- See science topics: Quantum Mechanics
Lecture:
The Changing Concept of the Atom
Once the concept of the atom as an indivisible particle was shattered and enough people accepted the view of the "component" atom, physicists were able to gain approval and grants to investigate this new view of matter further.
A Pictorial History of the "atom"
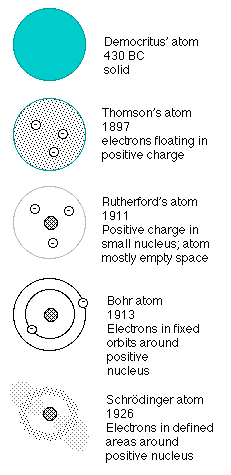
One of the problems facing the new physics of "quantum mechanics" was its deviation from the tried (but no longer quite true) principles of deterministic classical mechanics.
Read through Black Body Radiation at the University of Virginia Physics course site. You can skip the heavy math derivations and the section on degrees of freedom; concentrate on understanding the concepts!
- What is a blackbody?
- How does a body absorb light energy?
- How does a body radiate energy? How does the radiated energy differ from the absorbed energy?
- How does radiated energy depend on temperature?
- What was the ultraviolet catastrophe?
- How did Planck suggest the catastrophe be resolved? What observational or theoretical proof did he have at the time?
This period of physics recalls the medieval problem of Aristotelian cosmology, explaining matter with its solid spheres and laws of linear motion for earth, fire, water, air, and circular motion for the quintessence, but unable to predict the motion of the planets accurately, and Ptolemaic models that could predict the motions of the planets but had no justification from physical theory.
Read through One Hundred Years of Quantum Mechanics by Daniel Kleppner and Roman Jackiw. The first part recaps Planck's work, but the rest of the article summarizes the development of Quantum Mechanics from Planck's proposal.
- What was the dilemma of having electrons and protons in the same atom? How did Niels Bohr account for stable electron orbits?
- Follow the link for Wolfgang Pauli. What is the exclusion principle? How can this explain the periodic table?
- Use this link instead of the one in the article for Werner Heisenberg. What is the uncertainty principle?
- Follow the link for Erwin Schrödinger. What does his equation describe? How is it different from the descriptions of other moving objects?
- Follow the link for Paul Dirac. what is an antiparticle? Why did Dirac think antimatter existed?
- Follow the link fo rLuis-Victor de Broglie. What do we mean by the "wave nature of matter"? Why can't we see waves for large masses?
But how does this new view of the atom affect our understanding of chemistry and chemical compounds? Linus Pauling's ground-breaking work showed how electron orbital configurations can be used to predict molecular possibilities.
Read the first two pages of Linus Pauling's The Nature of the Chemical Bond.
- What types of chemical behavior and characteristics of chemical bonds does Pauling think his theory explains?
- When can two atoms form a stable molecule?
- When does an electron pair form? What are the two conditions placed on electrons in a pair?
Study/Discussion Questions:
- What objections did each new theory on the atom face?
- Why were these new theories not immediately accepted?
- What evidence finally convinced the scientists to accept it?
Further Study/On Your Own
- Explore the theory of Molecular Orbitals. This theory tries to explain molecules as if electrons were a common resource among all the atoms involved in each molecule, instead of forcing bonds to exist between two atoms only. Both the "two atom bond" or valence bond models and the molecular model are needed to explain different molecules. Neither theory explains all the molecules we know!
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.