
Natural Science - Year II
Unit 39: Search for the Elements

Science Web Assignment for Unit 39
| This Unit's | Homework Page | History Lecture | Science Lecture | Lab | Parents' Notes |
Science Lecture for Unit 39: Modern Chemistry and the Periodic Table
For Class
- Topic area: Chemistry
- Terms and concepts to know: Metals, acids, bases, non-metals, transition elements, alkaline metals, noble gases, diffusion, kinetic theory of gases, pH measurements, the ideal gas law.
- See historical period(s): 19th Century.
Outline/Summary
The Periodic Table
Units of measurement
Among of the tools chemists have had to develop were unit measurements for characteristics like volume, mass, pressure, and temperature.
- Units of volume, remember, are derived units, based on three dimensional measures of length. In modern science, the basic unit of length is the meter. A centimeter is 1/100 of a meter, a kilometer is 1000 meters. A cubic meter is a volume one meter on each side. More commonly used for liquid measurement is the liter, or one cubic decimeter ( 1/10 of a meter on each side), roughly equal to one quart.
- Mass is measured in grams, where a gram is the amount of water at 4 °C in 1 cubic centimeter ( = 1 milliliter or 1/1000 of a liter).
- Pressure is the amount of force exerted on an area, which is Newtons (remember that the unit of force was named for Sir Isaac) per meters-squared. More commonly used with gases, however, is the unit atmosphere, which is equal to the pressure exerted by the Earth's atmosphere at sea level, about 14.7 lbs. per square inch.
- Temperature is commonly measured using one of three different scales:
- Fahrenheit (32 °F = freezing, 212 °F = boiling point of water) for countries using British units, which includes the United States.
- Celsius (0 °C = freezing point of water, and 100 °C = the boiling point of water) for most scientific research.
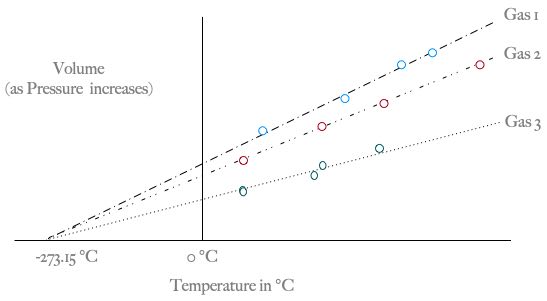
- The Kelvin scale is used when working with gas laws. The Kelvin scale has degrees which are the same size as the Celsius scale, but the zero point is at absolute zero, so 0K = -273.15 °C.
Having common units of measurement makes comparing experimental results and detecting patterns of behavior in chemical reactions much easier.
The Gas Laws
We have mentioned briefly several gas laws which were important in the quest for the nature of elements. To review
- Dalton's law of partial pressure means that the total pressure exerted by a mixture of gases on their common container is the sum of the individual pressures exerted by each gas. This law is very useful for analyzing mixtures. For example, if oxygen exerts 0.20 atmospheres and nitrogen exerts 0.79 atmospheres at the Earth's surface at 4 °C in a articular mixture of oxygen and nitrogen, the total pressure exerted by both gases is 0.99 atmospheres. Since the total pressure of air under these conditions is 1.00 atmospheres, there has to be at least one more gas (or group of gases) in air which makes up the missing 0.01 atmospheres of pressure. The actual group of gases includes CO2 and argon.
- Charles's law states that the volume of a gas at constant pressure depends only on its temperature: volume is proportional to temperature. We can write this mathematically as V = kT, where k is some constant of proportionality to make the units come out correctly.
- Boyle' law says that the volume of a gas at constant temperature depends only on the pressure, so V (holding T constant) = k/P
- The ideal gas law puts the laws about gas behavior together. It can be written as
PV = NkT
where P is pressure, V is volume, N is the number of moles, k is the proportionality constant (which is the same for all gases under all conditions) and T is temperature.
- If we plot the volume of a gas as a function of temperature for constant pressure, and extrapolate based on our experimental data, V goes to zero where T = -273.15 °C. We can't, obviously, push a gas to zero volume, so this is a theoretical "absolute zero", the coldest any substance could ever get.
Families of matter
Take a look at Mendeleev's early draft of the periodic table. In this version, we see eight columns. Elements in the first column on the left "Gruppe 1" or Group 1, combine in the ratio 2:1 with oxygen. You can see sodium (Na) and potassium (K) listed in column. Their placement here means they combine in this 2:1 ratio with oxygen, so the formula for sodium oxide would be Na2O and for potassium oxide would be K2O.
The next column, Gruppe II, combines in a 1:1 ratio with oxygen. For example, magnesium oxide is MnO.
The next column, Gruppe III, contains elements that combine in a 2:3 ratio with oxygen. So aluminum oxide is Al2O3.
- How does carbon in group IV combine with hydrogen? with oxygen?
- What does the number 23 in "Na = 23" mean?
- How are the elements arranged within each column?
Mendeleev's periodic table has been expanded and filled in; there are now 92 elements found in nature as well as a number of "trans-uranium" elements which can be produced in the lab and which have very short lifetimes before they decay into one of the more stable elements.
Look at the layout of a modern periodic table, and read about the different groups of elements.
- What are the characteristics of metals in general?
- What are the characteristics of the alkaline and earth alkaline metals?
- What are the characteristics of the transition metals?
- What are the characteristics of non-metals?
- What distinguishes halogens from other elements?
- What distinguishes noble gases from other elements?
Study/Discussion Questions
- What criteria are used to organize elements in the periodic table?
- What characteristics do elements in the same row share? In the same column?
- Why is the conservation of matter important to balancing chemical equations?
- What assumptions do we make in formulating the ideal gas law?
Further Study On your Own (Optional)
- Use your browser to find and examine at least three different representations of the periodic table. Why are they different? What does one include that the others do not? What pattern(s) does each emphasize?
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.