
Natural Science - Year II
Unit 37: Chemical Reactions

Science Web Assignment for Unit 37
| This Unit's | Homework Page | History Lecture | Science Lecture | Lab | Parents' Notes |
Science Lecture for Unit 37: Lavoisier and Chemical Reactions
For Class
- Topic area: Chemistry: Molecules and Bonds
- Terms and concepts to know: Ionic bonds, covalent bonds, molecules, lattices
- See historical period(s): 17th century - 19th century
Outline/Summary
Molecules and Bonds
We'll come back to the quest for individual types of atoms (elements) in our next unit, but for now, we want to look at how atoms combine into molecules and how these molecules interact to rearrange atoms into new combinations.
Molecules
Molecules are combinations of bits of elements (let's go ahead and call them atoms) that are bonded together. Today we can explain bonds as the result of electrical attractions between the positively-charged protons in the nucleus of one atom and the negatively charged electrons in orbit around the nucleus of another atom, but in Lavoisier's and Dalton's times, chemists simply recognized that different elements combined in discrete proportions; they didn't understand why these bonds exist or how they formed.
Right now, we have to skip ahead a bit. I realize that we haven't yet covered the historical events that led to the discovery of electrons, protons, and neutrons as parts of the atom...but that's one of the problems of studying the universe. Everything seems to be interconnected, and you need to learn some things out of order, and come back to fill in any holes as you learn more details. So for now, accept that atoms are not the "discrete, solid" particles that Democritus or even Dalton would describe, but that they have a few bits and pieces. We'll find out lots more about this when we get to the Curies and quantum mechanics, I promise!
Consider a water molecule: Dalton would describe it as something that had the proportions of 8 parts of oxygen to 1 part hydrogen by mass, or 2 parts of hydrogen to one part of oxygen by volume. We realize now that the volume measure gives us an idea of how many units (atoms) of oxygen and hydrogen are related, so the smallest particle that is "water" has two hydrogen atoms and one oxygen atom, combined together somehow and working as a unit. We can combine the same elements in different proportions to make a molecule of some other substance. If we combine hydrogen in 2 parts by volume to 2 parts of oxygen, we get a substance you know as hydrogen peroxide, the fizzy stuff you may put on a cut to sanitize a wound. Water and hydrogen peroxide are both combinations of oxygen and hydrogen, but because their proportions and molecular arrangements are different, they react very differently with other chemicals.

Covalent Bonds
Two major kinds of bonds hold atoms together in molecules. In covalent bonds, atoms share electrons. The negatively-charged electrons in one atom are attracted by the positively charged nucleus of the other atom, and this attraction is enough to hold the two atoms together.

Ionic Bonds
Ionic bonds occur when the attraction of one atom for the shared electrons overpowers the attraction of the other atom for its own electron. The atom with the strong "electronegative force" actually steals the electron from the weaker atom. Now the two atoms exist as ions with net charges. One atom has one more electron than it has protons, so its charge is negative, while the other atom has one less electron than it has protons, so its net charge is positive. We call these atoms ions to indicate that they have a net charge (cations have a positive charge, anions have a negative charge). Because opposite charges attract one another, the charge difference between the two ions creates an attraction that holds the atoms together.
The most common example of an ionic bond is table salt, NaCl. The sodium (Na) atom doesn't hang onto on of its electrons very well. Chlorine, on the other hand, has a "hole" for one electron in its own electron configuration, and is trying to find electrons to fill the hole. When Na and Cl come together, Na donates its outermost electron to the chlorine atom. Instead of sharing, chlorine takes the electron over entirely. But now, since the extra electron means that chlorine is negatively charged, while the lost electron means that sodium is positively charge, a difference in charges holds the two atoms together.
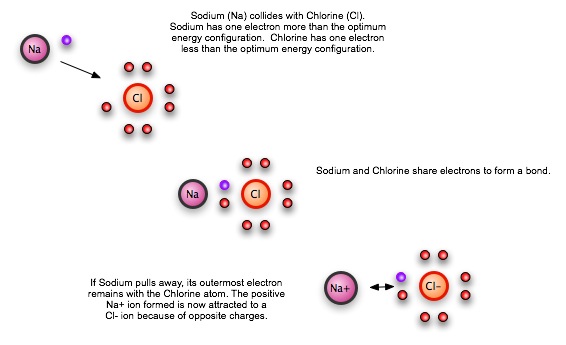
In general, ionic bonds are weaker than covalent bonds between atoms in molecules.
Lattices
Substances that form ionic bonds don't tend to form individual, isolated molecules, like water. Instead, they form complex lattices, where the pattern of attracted ions is repeated over and over until we run out of atoms. In this case, each Na+ ion attracts four chlorine atoms in its own plane, plus one in front and one in back. The ration is still 1 chlorine per 1 sodium, but there is no "molecule" that is contains one chlorine and one sodium atom.

Ionic substances are not the only type of matter that forms lattices. Substances can be allotropic arrangements, where all atoms are the same element. Most allotropic arrangements are lattices: instead of distinct and easily separated units like molecules, the arrangement can continue infinitely. Graphite, for example, is a form of carbon where the atoms form lattices that are 2-dimensional like a sheet of paper. The atoms in different layers are not connected by strong bonds, so sheets of graphite are easily detached from the main body. This is how your "lead" pencil works — it isn't lead at all, but a "sheet" form of carbon! In a diamond, each carbon atom connects to other carbon atoms in three dimensions. The atoms reinforce bonds — which makes diamonds one of the hardest substances known.

Most metals like copper and silver also form lattice arrangements. In metals, this means the electrons move more freely between atoms, and this flow of electrons forms electrical current, a way of moving energy easily from one place to another.
Intermolecular Bonds
Molecules do not directly share electrons with each other, but local areas of concentrated charge can create situations where one side of one molecule is positive, while the other side is negative. The local positive charge will attract local negative areas on other molecules. This causes the molecules to stick together in solid and liquid forms. Intermolecular forces between water molecules cause ice to form if the temperature is cold enough that individual water molecules slow down their normal motion and can orient themselves to form crystals. Even in liquid water, the attraction between water molecules creates surface tension strong enough to support small insects.

Study/Discussion Questions
- What is the common force that holds atoms together in molecules and molecules together in substances?
- How does the type of bond determine what kind of arrangment a molecule or lattice can have? What type of arrangements molecules in a substance can have?
- What do we mean by saying that chemistry is a study of "matter in different proportions"?
Further Study On your Own (Optional)
- Here is an animation explaining the difference between ionic and covalent bonds from the ZME science website.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.