Natural Science - Year II
Unit 36: Acids and Bases

Science Web Assignment for Unit 36
| This Unit's | Homework Page | History Lecture | Science Lecture | Lab | Parents' Notes |
Science Lecture for Unit 36: The Origins of Modern Chemistry
For Class
- Topic area: Chemistry: Acids and Bases
- Terms and concepts to know: Chemical property
- See historical period(s): 17th century - 19th century
Outline/Summary
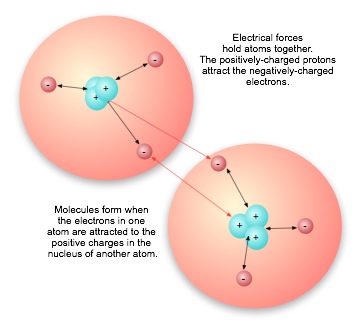
A refresher course on atoms
Click on the image of the two atoms to review basic properties of atoms and their interactions.
Acids and Bases
Elements and compounds
An element is a type of atom, a kind of matter which cannot be broken down any further without losing its unique characteristics. Atoms of the same element have the same chemical properties. They combine with other atoms of different elements in the same way. For example, an atom of carbon can combine with atoms of oxygen in one of two ways (CO, CO2), but not in other ratios.
A molecule is a particle made up of two or more atoms. The simplest molecules have two atoms of the same element. For example, oxygen occurs in our atmosphere in molecular form as O2. However, some organic molecules like proteins can have hundreds of atoms, and DNA strands are single molecules which have thousands of atoms.
A compound is a molecule with two or more elements represented. Water, H2O, is a compound.
States of matter
You should review the basic properties of matter we discussed in the second unit of this class, especially the definitions and properties of solids, liquids, and gases. For our purposes this week, we don't care that atoms can be broken down into subatomic particles like protons, neutrons, and electrons--that discovery comes later.
- Solids are composed of atoms or compounds of the same or different atoms (molecules) in fixed positions. Solids hold their shape, regardless of the type of container they occupy.
- Solids are often composed of crystals.
- Liquids are composed of atoms or molecules which are loosely connected and constantly change position. A liquid changes shape to fill the lowest parts of its container.
- Gases are made of individual atoms in constant motion, bouncing off of each other and all the sides of their container. Some special gases are monatomic, that is, they are made up of single atoms and no molecules, but most gases are made up of molecules.
- Gas particles move very quickly. The temperature of the gas is a measure of how fast the molecules or atoms of gas are moving.
- Gas particles exert pressure on the sides of their container.
- Gases have no definite volume--they expand to fill the container.
- Boyle's law states that the volume of the gas times its pressure is a constant for a given amount of gas at constant temperature: PV = k
- Charles's law says that volume varies inversely with temperature at a constant pressure: V/T = k
- Plasmas occur when there is enough energy under the right conditions to separate electrons from their atoms, forming free electron streams and positive ions. They occur in nature as the ion streams of the Aurora Borealis, and in stars. Man-made plasmas occur in fluorescent lights.
- Bose-Einstein Condensates are a recently-created form of matter, although Einstein predicted its possibility in the 1920s. Atoms in BE condensates are super-cold (just a millionth or billionth of a degree above absolute zero) and can pack tightly into clumps far more dense than the crystalline lattice formats of normal solids. Atoms this close tend to act almost exactly the same way, and become superconductors.
Physical Properties of matter
The physical properties of matter are those properties which belong to the substance itself and don't determine how the substance reacts with other kinds of matter. Physical properties include density, color, hardness, and taste.
Most kinds of matter exist as solid, liquid, or gas forms depending on the temperature. A physical change occurs when a type of matter moves from one phase to another. The temperature at which a substance changes from a solid to a liquid is its melting point; the temperature at which the substance changes from liquid to a gas i s its boiling point. For water, the melting point is 0 degrees Celsius (or 32 degrees Fahrenheit) and the boiling point is 100 degrees Celsius (212 degrees Fahrenheit). The amount of heat necessary for a given mass of solid to become liquid is called the substance's heat of fusion. The amount of heat necessary for the mass to become a gas is the substances heat of vaporization.
We can draw a phase diagram of most kinds of matter that looks like this:

Notice that while the solid is melting or the liquid is boiling away, the temperature of the substance does not rise. That is because all the heat energy is going into the phase change, and not into making the substance hotter.
Chemical Changes
A chemical change occurs when two chemicals react with one another. Remember Stahl's acid-alkaline experiments with baking soda and vinegar? When these two substances combine, they both change form to release carbon dioxide. The new substance has different properties from either of the original substances (for one thing, it's a gas at room temperature!).
A chemical change can be described by formula which specify the reactants going into the chemical reaction and the products coming out. We could write a rough approximation for this reaction as
vinegar + baking soda → carbon dioxide + other stuff + energy
We include energy as one of the products because we can see the bubbling effect of producing the carbon dioxide. Most chemical reactions do involve a change in energy as well as in the recombination of elements into different molecules.
Mixtures and solutions
Substances can be combined in physical mixtures, which can separated by physical means. For example, you can mix salt and sand together--the result is a mixture. Neither salt nor sand changes form or properties as a result of the mixture. How could you recover the salt and separate it out from the sand again?
Solutions are mixtures in which the space between one kind of atom or molecule is filled by another atom or molecule. A solution is composed of a solvent (whatever is present in the greatest quantity, or a liquid if the other components are not liquids) and one or more solutes that are dissolved in the solvent. Water acts as a solvent for table salt; when salt is put into water it forms a salt solution in which the solid salt crystals have dissolved.
It is possible to have solid solutions. Steel is a solution of carbon and iron.
It is also possible to have solutions where the solvent is liquid and the solute is a gas: your soda pop is an example of a solvent (the Coke or Pepsi syrup) containing carbon dioxide gas bubbles as the solute.
Obviously, it is possible to have gas "solutions": the atmosphere is a solution containing oxygen, nitrogen, carbon dioxide and traces of argon and other gases.
A solution is concentrated with there is a large amount of solute compare to solvent. A solution is saturated when the solvent contains so much solute that it can no longer dissolve any more solvent. If you have every made rock candy, you created a saturated sugar solution which, when it cooled, could no longer contain all the dissolved sugar, and so forced it out of solution into crystal form again.
Acids and Bases
Robert Boyle identified two kinds of chemicals: acids tasted sour, corrode metal, and become less acidic if mixed with bases. Bases feel slippery (think of detergent in water). Both change the color of "litmus", a lichen-based dye. Acids turn it red, bases turn it blue.
As with Kepler's laws, these observations reflected an empirical relationship, one that Boyle could observe over and over, but not explain. He just knew that some compounds fell into the "acid" category, and some fell into the "base" category. This pattern was so obvious that many different chemists have observed it under different circumstances. There are at least three different definitions for what an "acid" or a "base" is. They all depend on some concepts we haven't covered yet, but which you already may know: that atoms break down into parts, and those parts include "subatomic particles" like protons (with a positive electrical charge) and electrons (with a negative electrical charge). We'll talk more about how these fit into an atom, but for now, with regard to acids and bases:
- Arrhenius definition: an acid is a something that increases the concentration hydrogen radical in water; bases reduce it.
- Bronsted-Lowry definition: an acid donates a proton, a base accepts one.
- Lewis definition: an acid accepts a pair of electrons; a base donates them.
Read the introduction to acids and bases at Visionlearning's Acids and Bases site.
- Note the pictures of Arrhenius and Bronsted -- you can read about each one but don't have to for this lesson.
- What is a hydrogen ion?
- What is pH? how do we use it to measure acid strength?
- Which of the following are strong acids: HCl Vinegar Rainwater
- Which of the following are strong bases: Baking soda, ammonia, lye (NaOH)
- What happens when you combine a strong acid with a strong base?
The key point to notice here is that acids and bases react in pairs: we have acid-base reactions, not acid reactions alone. There are other reactions that don't fall into one of the three categories above, but when a reaction does fall into an "acid-base" pattern we can make some assumptions about what is happening.
Because acid-base reactions involve the transfer of charged particles, they are important to our understanding of electrical current. We'll come back to this later.
Study/Discussion Questions
- Physical properties involve characteristics that involve changes of state but not composition, which is a chemical change. If you were given a new situation to analyze, what characteristics would you look at to determine whether a change is a physical or chemical change?
- Elements, compounds, molecules, mixtures, and solutions are all different levels of organization of matter. How are they related?
Further Study On your Own (Optional)
- Read about phases of matter and the different types of chemical reactions at the The Chem Tutor.
- Read about Bose-Einsteian condensate at the NASA website.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.