
Chemistry
Chemistry 8: 4-6

WebLecture
Electron Bonds and Resonance
Outline
Single and Multiple Bonds
In covalent bonds, two or more atoms share electrons, but that doesn't mean a given atom shares all its electrons. Atoms that have paired electrons can't share the completed pairs; they can only share unpaired electrons. We use Lewis dot diagrams to help analyze molecular bonds between atoms.
Electron Pairs
Consider the molecule SO2.
1. The first thing we do is determine the arrangement of valence electrons around each type of atom when it is by itself. Oxygen has the configuration 1s2 2s2 2p4, so there are six valence electrons for each atom.

2. Sulfur has the configuration 1s2 2s2 2p6 3s2 3p4, so it has six valence electrons also.

3. We now arrange the two oxygens around the central sulfur in such a way that the sulfur atom has single bonds with each oxygen.

4. Each oxygen atom now has a complete octet, but the sulfur does not. If we move one of the electron pairs on one of the oxygens around to form a double bond with the sulfur, all three atoms will have complete octets.

5. The leftmost oxygen has three pairs of electrons that are not part of bonds, the rightmost oxygen has two nonbonding electron pairs. These lone pairs are important; they will eventually help us figure out the shape of molecules.
There are exceptions to the octet rule. BCl3 has fewer than 8 electrons around the central atom, SF6 has more than 8. Radicals such as NO have an odd number of valence electrons, so not all of them can be paired. These molecules are generally reactive, forming bonds with other atoms to achieve octet status.
Multiple Bonds
In the above analysis, we had to double up electron bonds to create an octet for sulfur. Multiple bonds are quite common, since they allow a central atom to form molecules with fewer other atoms and still complete an octet. When more electrons take part on a bond, the bond becomes stronger. Oxygen gas forms a double bond O2, but nitrogen gas forms a triple bond, which is extremely difficult to break without an appropriate catalyst. For this reason, while N2 forms over 78% of the earth's atmosphere, it does not react with many substances, including hydrogen, except where bacteria possessing appropriate enzymes can "fix" the nitrogen to hydrogen to make ammonium ions, NH4+.
Atoms can form double or triple bonds; they do not form quadruple bonds, for reasons we will look at in the next chapter.
Resonance
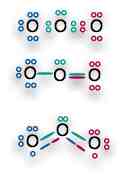
In our SO2 molecule, it doesn't matter which sulfur forms the double bond, either originally, or from time to time. The two bonds have an equal quantum possibility, so the electrons cannot "chose" one over the other.
Resonance structures are particular common in organic compounds, especially in chains or rings of carbon atoms, such as the benzene ring formed. The resonance bond is a hybrid of the two possible bond situations. [For a good discussion on how to draw resonance bonds using Lewis diagrams, see Mark Bishop's Chemistry Site: Resonance web page.]
The textbook explains how to represent resonance, but representations don't describe what is actually happening in a resonance situation. We do not have two stable bonds, one single (longer and weaker) and one double (shorter and stronger), that rapidly switch places. This would give us an extremely unstable molecule. Experimentally, all evidence indicates that the "double" and "single" bonds in resonance molecules are of equal length and strength.
We can only explain this if the "extra" bond in a resonance situation is delocalized, with its electrons smeared across a hybrid area that covers both bonds. Rather than having a particle electron hopping back and forth between two states, unable to make up its mind where to stay, we have a special "orbital" state in which the resonance electron is actually shared between three (or more) atoms. We can represent this hybrid orbital using a dashed-line for the resonance hybrid.
Practice with the Concepts
Discussion Questions
- Why does sulfur not have a preference for which oxygen to use in forming the required double bond in SO2?
Optional Readings
- To learn more about drawing resonance structures, look at the UCLA Chemistry Department's Resonance Structures page.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.